A “Poor” PLM Microscopist’s Tunable Monochromatic Light Source
When doing some more sophisticated work with a polarized light microscope (PLM), from time to time the desire emerges to have a monochromatic light source at hand that is tunable over the entire visible light spectrum. One way of studying wavelength dependent phenomena in microscopy is the use of microspectroscopes or even microspectrophotometers, or, in the home shop, use the microspectroscopic eyepiece described in this journal by Delly [1]. For microscopists like me, doing most of their microscopy work in the home shop, the desire to have a tunable monochromatic light available is not so easily realized. Professional monochromators that are adapted specifically for use with a PLM are expensive. Classical tunable interference filters are difficult to obtain and are expensive, too. And even the single wavelength interference filters are only affordable for the most common wavelengths, like the Fraunhofer C, D, and F lines, if you can buy them as used items; e. g., through online auctions. This uncomfortable situation led me to the idea of building a “poor” PLM microscopist’s tunable monochromatic light source out of a used single-beam near-UV-VIS spectrophotometer and some cheap additional parts.
The PLM that I use in my home shop is an AMPLIVAL pol u from the seventies of the last century, made by Carl Zeiss, Jena. At that time, it was one of the leading instruments (if not the leading instrument) for polarized light microscopy available. It is a universal research grade transmitted and reflected light PLM, with many accessories not found so often these days, including (but not limited to) a four-axis universal stage, Wright eyepiece, rotatable polarizer and analyzer, a whole bunch of compensators, half shadow plates et cetera; see Figure 1 for a front view of this instrument.

The near-UV-VIS spectrophotometer that I use for the purposes described here was manufactured by the same firm: Carl Zeiss, Jena. It is a SPEKOL 11 single beam instrument, with different lamps in different housings attachable, with a manual wavelength selection drum on the front and (most important) a light port on its front plate which serves as a docking socket for external attachments, e. g. for cuvette holders of different sizes, and additional equipment. This leads to the not-so-common advantage that you (as the “poor” microscopist) have access to a perpendicular beam of monochromatic light emanating from the exit slit; see Figure 2.

Some of the optical characteristics of the monochromator used in the SPEKOL 11 can be found at the end of this article. Before I started with the setup of the combination of these two instruments, I made some preliminary tests on the geometry of the light beam that emerges from the light port/exit slit on the front plate of the SPEKOL 11. I used some glassine paper, glued on a microscope slide, as a little screen and moved this in the direction of light travel. It was easily observable that there is a focal point of the emerging light, approximately 20 mm away from the exit slit of the photometer, and then the beam opens up quickly. The next step was to check the same with the cuvette holder EK 1 attached and the optical system of this cuvette holder in place (see the black bakelite attachment screwed to the main unit of the SPEKOL 11 with two white knobs, left side of the instrument, just beneath the Name SPEKOL 11, in Figure 2, but with the photocell housing detached). This time, the geometry of the emerging light beam seems more appropriate for using it as an illuminating source in microscopy. The angle of the light cone was much smaller, so the loss of light intensity should be reasonably lesser than with the first setup. The final arrangement of components can be seen in Figure 3.

The AMPLIVAL pol u PLM has a built-in lamp for transmitted light microscopy. The design of this lamp enables full Köhler capabilities for the illumination of the specimen. In the baseplate of the instrument, there is a round port for filter glasses, and swing-in/out lens for the illumination of large object fields. On top of this swing-in/out lens, I fixed a tiltable plane mirror, projected the light coming from the spectrophotometer that stands to the left side of the microscope at right angles onto the plane surface of the mirror, and then adjusted the mirror in such a way that I can see a monochromatic band of light when looking in the eyepiece. This was then centered to coincide with the horizontal (or E-W) hair of the crosshairs. The image that you see through the eyepieces is shown in Figure 4, taken by the method of ocular projection.

If you prefer to have a fully illuminated field of view for observation and measurement, there are some additional tricks of the trade necessary to accomplish this. By using a cylinder lens, it is possible to spread out the horizontally running light beam into a field-filling cone of light. If you do not have a cylinder lens at hand like me (remember, you and I are the “poor” PLM microscopists), use a simple cylindrical drinking glass from your kitchen, fill it with water, and move it into the light path between the exit lens of the cuvette holder and the tiltable mirror. You will see that the field of view becomes almost uniformly illuminated, but light intensity is reduced, of course. Fix that drinking glass in this position, and you can manipulate your preparation, the rotating stage, and the fine focus with ease. (If you are interested in some other nice experiments with cylinder lenses and are able to read German, please visit the internet for reference [2]). [Another possibility to achieve this evenly illuminated field of view might be the use of a round bottom Florentine or Florence flask (Schusterkugel in German). Personal communication from John G. Delly, McCrone Associates].
At this point, I think it is wise to make some additional remarks on the optical and mechanical properties of the AMPLIVAL pol scope. This PLM is the only one that I have used so far that has an optical system which gives upright and left-right-corrected images. If you move your specimen to the right, the image moves to the right, too. If you move the slide upward, the image moves upward. This is achieved by a unique optical system above the analyzer and the stand, that also contains the focusable Amici-Bertrand lens and a special diaphragm to select the most interesting part of your specimen for observation in the conoscopic view. This special optical system also contains the crosshairs (they are not located inside of one of the eyepieces!). The factory setup of the polarizing equipment of the AMPLIVAL pol u is done in a way that the substage polarizer has its transmission direction North-South and the analyzer above the compensator slot has its normal position East-West when using them in the common crossed polars arrangement, which is opposite to the direction in the modern design of most PLMs. This is because the AMPLIVAL dates back in its optical design to the times before the arrangement of optical components in PLMs was standardized in what is nowadays DIN ISO 8576. This deviation in optical arrangement is not critical for most applications, but not for all; some more specialized techniques are sensitive to the arrangement of polarizing filters in the optical path of the instrument, e. g. the Herzog test to differentiate flax from hemp in fiber microscopy (see [3] for details). [If you are interested in a full description of the AMPLIVAL pol u, as well as many other microscopes and instruments from Carl Zeiss, former East and former West Germany (Jena and Oberkochen), please visit the vast collection of documents at www.mikroskope-online.de, then go to “Zeiss-Jena BDA” (in German only)].
In the applications described here, it would be favorable to have the privileged direction of the substage polarizer also East-West, because there you have the beam of monochromatic light, shining like a bar, coming from the above explained setup. Fortunately, the substage polarizer of this instrument is rotatable and can easily be moved by 90 degrees. To check for correct alignment in this “not notched-position,” it is advisable to use a test specimen (for instance, a very straight, man-made synthetic fiber, some crystals of natrolite or some long, recrystallized needles of lead chloride) and adjust the polarizer perfectly East-West, coinciding with the East-West hair (for details on the alignment of optical components of a PLM, see, for instance, the works of Puhan, Johannsen, McCrone, or Chamot and Mason). Last but not least, don’t forget to rotate the analyzer too (if you want to work with crossed polars…) and adjust this filter for complete extinction.
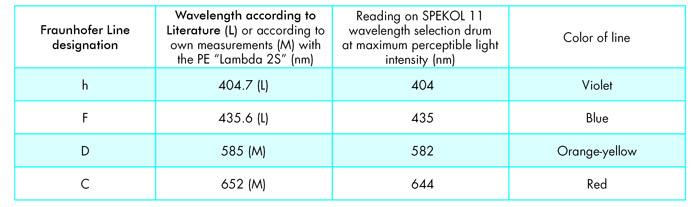
As with every self-constructed system, you should perform at least some rudimentary tests of the performance characteristics of your setup before starting with real experiments. One of my concerns was the trueness of the wavelength scale on the manual wavelength selector drum of the spectrophotometer, because this instrument had seen some long years of heavy-duty use in a routine analytical chemistry lab and has already some hard-earned stains and scratches on the outside. To check wavelength trueness with the “poor” microscopist’s limited means, I got some inspiration from Delly’s work [1] and decided to use an ordinary fluorescent light bulb instead of the halogen lamp as the light source for the SPEKOL 11. In the radiation emitted by these devices, you can see the spectral lines of the chemical element mercury as very sharp signals when looking at the bulb with a handheld straight-view spectroscope of the Browning type. The two mercury lines in the violet and blue region at 404.7nm and 435.6 nm can be seen as perfectly sharp signals. Unfortunately, in the turquoise, green, yellow, orange and red region of the light from my fluorescent light bulb, there were bands of light instead of sharp lines. This is the result of the phosphorescent layers on the inner side of the glass of these bulbs which are designed to transform the line spectrum of mercury to a desired wavelength “continuum.” So for the medium and longer wavelengths of the visible range, I used two commercially available interference filters as calibration standards. I checked their actual wavelength for the transmission maximum with a professional UV-VIS-Spectrophotometer Perkin-Elmer Lambda 2S.
The wavelength trueness tests with the SPEKOL 11 were performed in such a way that I manually tuned the wavelength drum, beginning in the near ultraviolet, while illuminating the entrance lens of the instrument with the light of the fluorescent bulb. When I saw maximum light intensity with my eyes on the little lamp-adjustment-screen made from ground glass, sitting on the light exit socket of the instrument, I read the wavelength at the indicator of the λ-drum. For the two interference filters, I used an external microscope illuminator with a tungsten-halogen lamp as the light source, focused the beam of light onto the entrance lens of the SPEKOL 11, and put the interference filters one by one between the lamp and the entrance lens at right angles to the optical axis of the setup to avoid wavelength shift because of not-normal light incidence on the dielectric layers of these filters. You can see the results of these wavelength trueness tests for the SPEKOL 11 in Table 1:
These data, taking into account the spectral bandwidth of the instrument, convinced me once again about the exceptional mechanical and optical performance characteristics of equipment made by Carl Zeiss even after decades of heavy-duty use. I decided that these accuracies are good enough for my home shop use of the setup.
After this setup and the tests, you now have a lot of possibilities for doing exciting experiments:
- Measure refractive indices of fibers, particles, et cetera at different wavelengths from 400 nm (approx.) to 700 nm (approx.), e. g. by the Becke line method, and construct complete dispersion curves.
- λ-T-variation methods according to R. C. Emmons for refractive index measurements, (Emmons double variation method), if you do have a hotstage for your microscope.
- Use tunable monochromatic light to study some more sophisticated effects in conoscopy, such as change of optic sign with wavelength change.
- Use monochromatic light for some applications in gemmology, where the wavelength-dependant absorption characteristics of some gemstones, e.g. emerald, are very characteristic (for details, see, e. g. [4]), and you can use these characteristics on small gemstone fragments under the microscope, too.
- Measure the dispersion of birefringence (see, e. g. [5] for details).
- Use monochromatic light for some applications in pigment microscopy where this can also be used to the fortune of the observer (for details, see, e. g. [6] and [7]).
- And there are innumerable other possible applications of this little attachment to the PLM. In my opinion, it is worth a study of the older literature (like the works of Johannsen, Berek, Burri) to find inspiration about what is possible with this setup.
Some practical (and legal) advice, at last:
The author does not take any responsibility with respect to the use of the equipment in the way described here; you are solely at your own risk when using equipment of this type in this manner.
The tunable monochromatic light source, as described herein, is intended for a hobbyist’s use, not for use in a professional surrounding/analytical laboratory, et cetera.
Equipment and Materials Used
Polarized Light Microscope AMPLIVAL pol u, Carl Zeiss, Jena, ca. 1970
UV-VIS Spectrophotometer SPEKOL 11, Carl Zeiss, Jena, ca. 1980-85
Some technical data on this instrument:
- Single beam near-UV-VIS-Spectrophotometer, Tungsten-Halogen-Lamp as standard light source, reflecting diffraction grating with 651 grooves/mm
- Wavelength range: 340 nm to 850 nm
- Reproducibility of wavelength selection: ± 0.2 nm
- Spectral bandwidth: 11 nm
- Fixed exit slit width design
- Browning type handheld spectroscope with straight sight, Carl Zeiss, Jena
- Interference Filters SIF 656 and SIF 589, for Fraunhofer Lines C and D, Carl Zeiss, Jena (SIF is an acronym for schmalbandiges Interferenzfilter)
Digital photographs shot with CANON EOS 350D digital, objective: Canon Zoom Lens EF-S 18-55 mm, 1:3.5-5.6 II, copyright for all photographs: Jan Burmeister, 2009.
Cited References
- Delly, John G. How to Make and Use a Simple Microspectroscopic Eyepiece. Accessed April 5, 2009
- Simple experiments with cylinder lenses, in German: http://marvin.sn.schule.de/~physik/optik/o2.php, accessed: April 11, 2009.
- Valascovic, G. A. Polarized Light in Multiple Birefringent Domains: A Study of the Herzog Effect. The Microscope, 39 (1991), 269-286
- Henn, Ulrich. Gemmologische Tabellen. Deutsche Gemmologische Gesellschaft, Idar-Oberstein, (no date)
- Beyer, H.; Riesenberg, H. (Eds.): Handbuch der Mikroskopie. 3rd ed., VEB Verlag Technik, Berlin 1988
- McCrone, Walter C. The Microscopical Identification of Artists’ Pigments. Journal of the International Institute of
Conservation, Canadian Group, 7 (1982), Nos. 1 & 2, pages 11-34 - Mactaggart, Peter & Ann. A Pigment Microscopist’s Notebook. Published by the authors, Higher Hare Farm, Chard, Somerset, UK 1998
General References and Reading Suggestions
Johannsen, Albert. Manual of Petrographic Methods. McGraw-Hill, New York 1914 (reprint in paperback: Kessinger Publishing, reprint not dated).
Berek, Max. Anleitung zu optischen Untersuchungen mit dem Polarisationsmikroskop. E. Schweizerbart’sche Verlagsbuchhandlung, 2. Auflage, Stuttgart 1953.
Burri, Conrad. Das Polarisationsmikroskop. Verlag Birkhäuser, Basel 1950.
McCrone, Walter C. Particle Characterization By PLM. Part III: Crossed Polars. The Microscope, 31 (1983) 187-206.
Herzog, Alois. Mikrophotographischer Atlas der technisch wichtigen Pflanzenfasern. 2. Auflage, Akademie-Verlag, Berlin 1955.
Chamot, Emile M.; Mason, Clyde W. Handbook of Chemical Microscopy. Two Volumes. Second Edition, John Wiley & Sons, New York 1954.
Emmons, R. C. The Double Variation Method of Refractive Index Determination. Amer. Mineral. 14 (1929) 414.
Puhan, Detlef. Anleitung zur Dünnschliffmikroskopie. Ferdinand Enke Verlag, Stuttgart 1994.
Contact the Author
Jan Burmeister
Mendelssohnstrasse 40
D-06844 Dessau-Rosslau
Germany
jan.burmeister@online.de
Comments
add comment